|
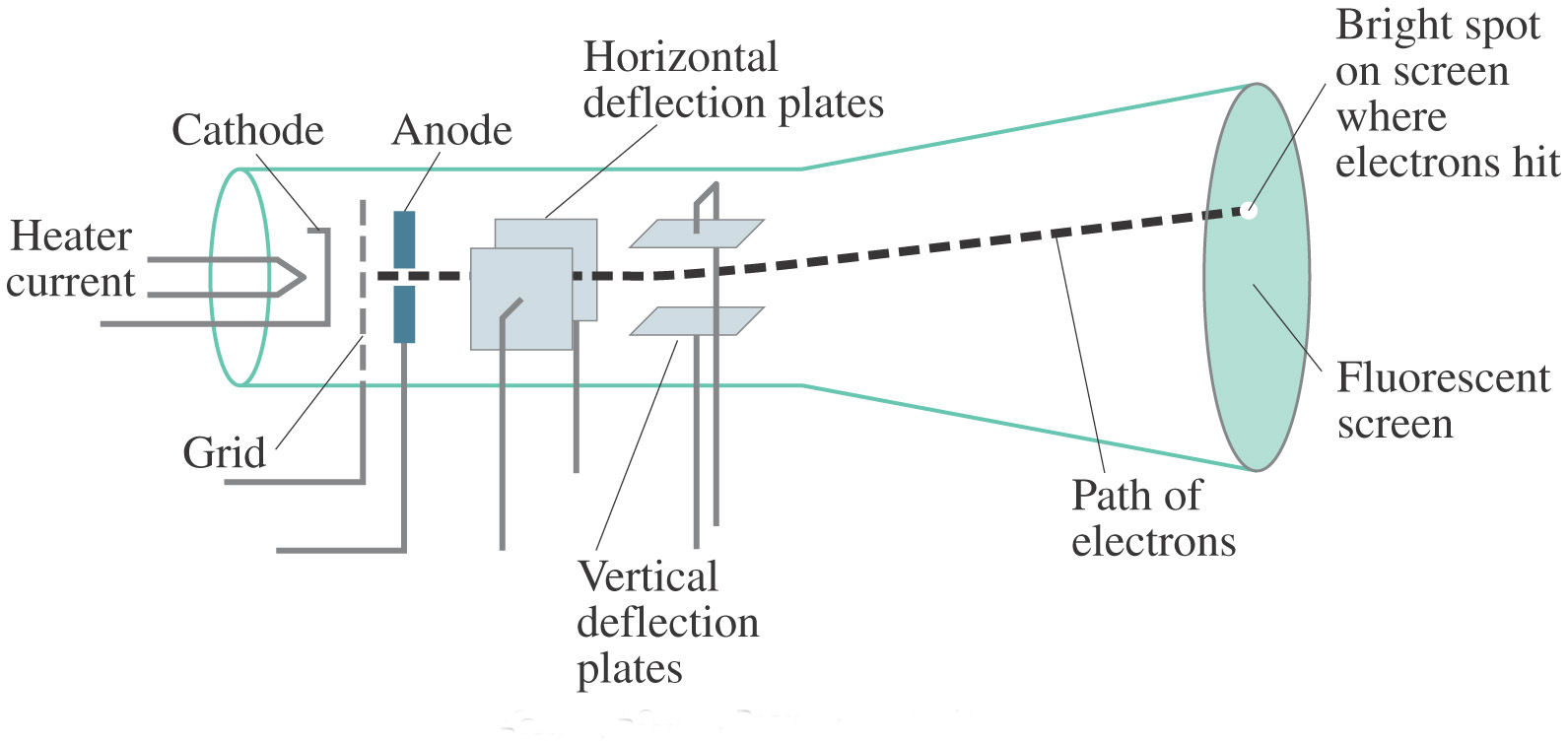
What are the key takeaways from the cathode ray tube experiment? (ii) At anode: Anode decreases in size due to the formation of copper ions. What is the observation at the cathode?Īt cathode: Greyish white metal lead is formed on the cathode. Using this information Thomson determined the mass to charge ratio of an electron. Then Thomson measured how much various strengths of magnetic fields bent the particles. Thomson was able to deflect the cathode ray towards a positively charged plate deduce that the particles in the beam were negatively charged. Thomson also concluded that electrons are part of atoms. In 1897 he showed that cathode rays (radiation emitted when a voltage is applied between two metal plates inside a glass tube filled with low-pressure gas) consist of particles- electrons-that conduct electricity. See also What Number Of Fundamental Particles Exist What two things did J.J. Thomson took science to new heights with his 1897 discovery of the electron – the first subatomic particle….Atoms 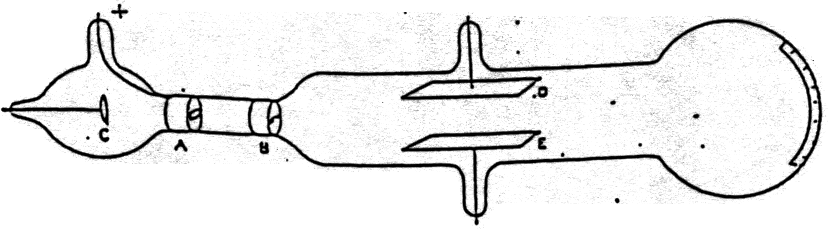
This device used a cathode ray tube to produce luminescence on a chemically treated screen. Practical Uses for Cathode Rays The first such application came in 1897 in the form of Karl Ferdinand Braun’s oscilloscope. What was the first practical purpose of the cathode ray tube? Though Thompson referred to them as “corpuscles,” what he found is more commonly known today as the electron. This finding revolutionized the way scientists thought about the atom and had major ramifications for the field of physics. Thomson announced his discovery that atoms were made up of smaller components. What was so significant about Thomson’s discovery? The tube contains an electron-gun structure (to provide a narrow beam of electrons) and a phosphor screen. The function of the cathode-ray tube is to convert an electrical signal into a visual display. Thomson discovered that the mass of the particles in cathode rays was 1800 times lighter than hydrogen, the lightest element. History of Cathode Rays Johann Hittorf discovered cathode rays in 1869 using a Crookes tube and noting shadows cast on the glowing wall of the tube opposite of the cathode. What cathode rays are and how they were discovered? What evidence from the Cathode Ray Tube Experiment support the claim that electrons have a negative charge? The Cathode Ray bent way from a negatively charged plate. See also What is the largest asteroid ever recorded? What evidence is found in the cathode ray tube? In addition, he also studied positively charged particles in neon gas. 
He demonstrated that cathode rays were negatively charged. Thomson discovered the electron by experimenting with a Crookes, or cathode ray, tube. Thomson, led to the discovery of the negatively charged part of the atom, the electron. The cathode ray tube experiment, originally carried out by J.J. Thomson proposed the plum pudding model of the atom, which had negatively-charged electrons embedded within a positively-charged “soup.” Why was the discovery of cathode rays important? Thomson’s experiments with cathode ray tubes showed that all atoms contain tiny negatively charged subatomic particles or electrons. What was discovered in the cathode ray experiment?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
